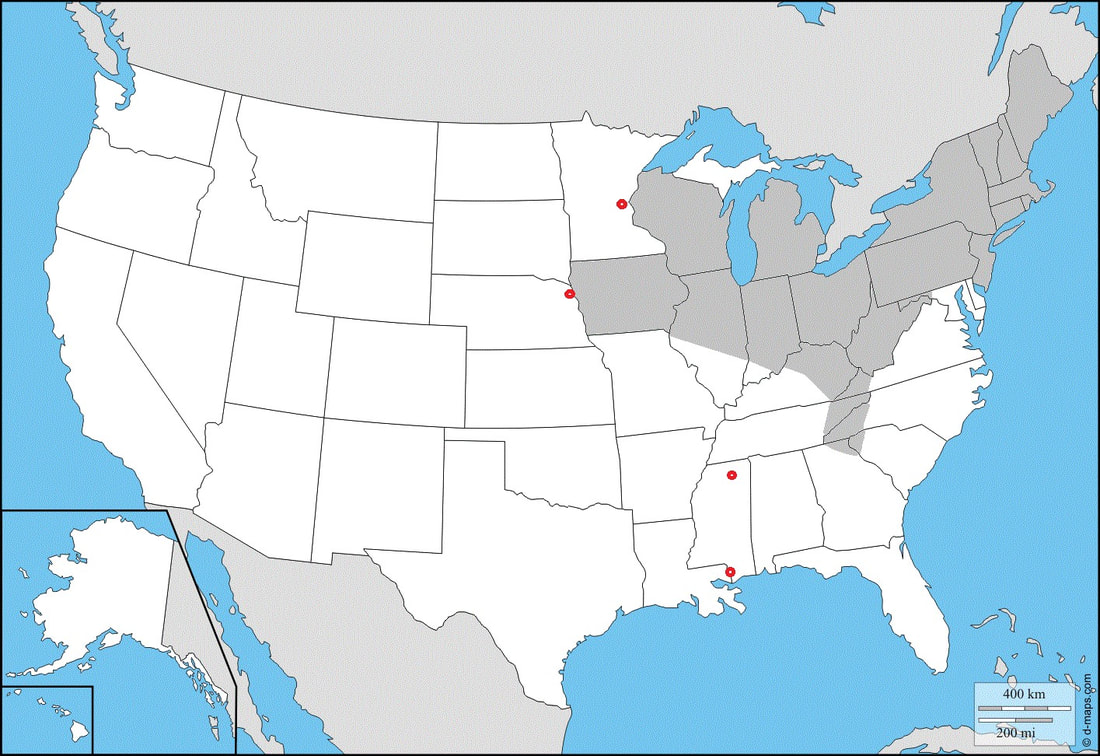
|
Common Name(s): Hermit Sphinx
Ecology and Life History: This moth flies in late June and July primarily. Given that it is not found very far south, it can not be found year-round anywhere in its range. This species will come to light, but never commonly. Bait is an ineffective method for trapping this species. Males and females are not dimorphic and have the same pattern. Females are slightly larger than males, and have a rounder abdomen. Eggs are laid on the leaves and stems of hostplants. This species prefers plants in the Lamiaceae (Mint Family) and as such can be a common garden visitor. The younger larvae tend to hide on the undersides of leaves near the stem, seeming to mimic the petiole of the leaf. Older larvae are more conspicuous and likely hide off the plant in dead leaves during the day. Habitat and Searching for Larvae: Searching plants in the Lamiaceae is the best way to start looking for this species. They have been found on Salvia, Agastache, Mentha, and Monarda. As none of these plants ever grow very tall, the larvae are never out of reach. Younger instars hide on the undersides of leaves near the stems, or sitting on the stem right underneath a leaf. At night, larvae ascend the plant and begin consuming leaves from the edge inward. They can often be found near the flowers feeding at night on some plants like Monarda. This species inhabits parks, edges, yards, gardens, fields, and wetlands where its hostplants grow. Larvae can be found from late July through September and October. Younger instars of this species fluoresce highly under UV light. Final instar larvae do not fluoresce. Rearing Notes: We have not been able to gather eggs from this species yet. Placing females in a paper bag or styrofoam cooler would likely work quite well. Alternatively, a flight cage with hostplants may work too. The adults do feed, and would likely need to be fed in captivity. These larvae accept many species in the Lamiaceae and are not picky, switching to various plants throughout their growth if offered. Larvae tend to grow quickly and largest on Monarda. This species is highly susceptible to disease. Occasionally large batches of larvae will all perish due to a viral or bacterial infection. Keep rearing conditions extremely clean, and not over-crowded will usually yield pupae. This species is sensitive to being crowded, it is not recommended to keep more than 8 individuals together when rearing in tupperware. More larvae can be housed together if using a large cage, or sleeve. Ventilation is key to rearing this species. Sleeving is a great method, and many larvae can be reared this way successfully. Pupation is simple using the paper towel method (outlined in the general information tab of this website) or allowing larvae to tunnel into a loose soil media. Host plants: Click here to load this Caspio Cloud Database
Cloud Database by Caspio |
Adult description:
This is a fairly small moth with forewings 27-37mm in length (1). The overall appearance of this moth is yellow-brown or gray and quite mottled. There are black and white streaks throughout the wings. There is a large silvery discal spot, and another smaller spot above it. The thorax is gray with various black lines. The hindwings are black with a white area near the top, and then a very distinctive white line in the middle of the wing. This moth can be confused with Paratrea plebeja and Sphinx canadensis. The hindwings are dramatically different than the solid black of Paratrea plebeja, and Sphinx canadensis lacks the discal spots. Larval description: L5: The larva is very distinctive. It is predominantly black and has a large black oval outlined by cream on the dorsal portion of the thoracic segments, usually right behind a raised area. The horn is small, black, and hooks sharply backward. The spiracles are well defined and outlined with black circles. There are usually white lines on the abdominal segments sometimes bisecting the spiracles. There are cream speckles on the larva. |
The gallery to the left contains photos of Lintneria eremitus adults. If you have a photo that you would like to submit to us, please contact us.
The gallery to the right contains photos of Lintneria eremitus larval and pupal stages. If you have a photo that you would like to submit to us, please contact us.
The gallery to the right contains photos of Lintneria eremitus larval and pupal stages. If you have a photo that you would like to submit to us, please contact us.
|
|